How Bleach is made
The manufacturing process of sodium hypochlorite bleach is lengthy, but everything can be done at one large manufacturing facility.
Raw Materials
The raw materials used for making bleach are chlorine, caustic soda, and water.
The raw materials used for making bleach are chlorine, caustic soda, and water.
Resource Extraction
Chlorine and caustic soda are produced by putting electricity through a sodium chloride salt solution. This process is called electrolysis. Sodium chloride, commonly known as table salt, comes from either mines or underground wells. The salt dissolves in hot water to form a salt solution which would be tested for impurities before the electrolysis process ("How Bleach is Made", 2007).
Chlorine and caustic soda are produced by putting electricity through a sodium chloride salt solution. This process is called electrolysis. Sodium chloride, commonly known as table salt, comes from either mines or underground wells. The salt dissolves in hot water to form a salt solution which would be tested for impurities before the electrolysis process ("How Bleach is Made", 2007).
Materials Processing
Obtaining the Materials
Chlorine is made in liquid form outside the manufacturing facility, and brought to the facility through specially built tank cars with double walls that will not break in case of a derailment. Once the liquid chlorine arrives at the plant, it is pumped from the tank cars into holding vats (tanks used in factories to hold liquids). As a safety precaution, the tank cars also have shutoff valves that stop pumping chlorine if the chlorine detection system goes off. This way, if there was a chlorine leak, it will automatically stop the leak in 30 seconds. Once the chlorine vats are inside the facility, they're put in an enclosed area called a car barn. The car barn contains "air scrubbers", which clean the air of any escaped chlorine gas that can harm humans and the environment. The chlorine gas is later used to react with the caustic soda to create bleach ("How Bleach is Made", 2007).
Preparing the Components
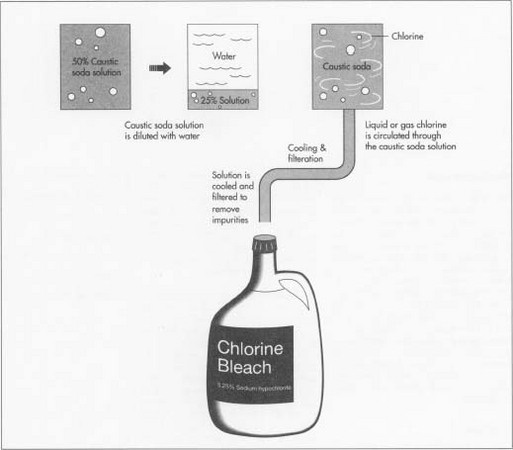
Caustic soda is produced as a concentrated 50% solution, then shipped. At the manufacturing facility, the solution is diluted with water to create a new 25% solution. Since heat is created when the water dilutes the solution, it is cooled before its reaction ("How Bleach is Made", 2007).
The Chemical Reaction
Chlorine and the new caustic soda solution is reacted, which creates sodium hypochlorite. The chlorine is circulated through the caustic soda. The reaction will happen instantly, and it can take place in a batch of 14,000 gallons, or can be done in a continuous reactor ("How Bleach is Made", 2007).
Cooling and Purifying
The bleach is cooled to prevent decomposition, and then it is put through a series of filters to remove impurities that can discolor, decompose or do anything else to the bleach. It is also tested to make sure it contains exactly 5.25% sodium hypochlorite. Safety is a great concern at the manufacturing facilities because of the presence of unstable chemicals ("How Bleach is Made", 2007).
Obtaining the Materials
Chlorine is made in liquid form outside the manufacturing facility, and brought to the facility through specially built tank cars with double walls that will not break in case of a derailment. Once the liquid chlorine arrives at the plant, it is pumped from the tank cars into holding vats (tanks used in factories to hold liquids). As a safety precaution, the tank cars also have shutoff valves that stop pumping chlorine if the chlorine detection system goes off. This way, if there was a chlorine leak, it will automatically stop the leak in 30 seconds. Once the chlorine vats are inside the facility, they're put in an enclosed area called a car barn. The car barn contains "air scrubbers", which clean the air of any escaped chlorine gas that can harm humans and the environment. The chlorine gas is later used to react with the caustic soda to create bleach ("How Bleach is Made", 2007).
Preparing the Components
Caustic soda is produced as a concentrated 50% solution, then shipped. At the manufacturing facility, the solution is diluted with water to create a new 25% solution. Since heat is created when the water dilutes the solution, it is cooled before its reaction ("How Bleach is Made", 2007).
The Chemical Reaction
Chlorine and the new caustic soda solution is reacted, which creates sodium hypochlorite. The chlorine is circulated through the caustic soda. The reaction will happen instantly, and it can take place in a batch of 14,000 gallons, or can be done in a continuous reactor ("How Bleach is Made", 2007).
Cooling and Purifying
The bleach is cooled to prevent decomposition, and then it is put through a series of filters to remove impurities that can discolor, decompose or do anything else to the bleach. It is also tested to make sure it contains exactly 5.25% sodium hypochlorite. Safety is a great concern at the manufacturing facilities because of the presence of unstable chemicals ("How Bleach is Made", 2007).
Packaging
Sodium hypochlorite bleach was first introduced to Americans in 1909 in steel containers. It was later sold in glass bottles, but it wasn't until the early 1960's, that the plastic jug was introduced. This cheaper, lighter, and non-breakable packaging alternative reduced transportation costs, provided safety to the workers involved in shipping and handling, and also prohibited ultraviolet (UV) light from reaching the bleach, which improved its chemical stability and effectiveness. However, in recent years this plastic became an environmental issue, so now the amount of plastic used to create the packaging is reduced, and more recycled plastic is used ("How Bleach is Made", 2007).
This video shows how bleach is filled and capped into plastic bottles.
Sodium hypochlorite bleach was first introduced to Americans in 1909 in steel containers. It was later sold in glass bottles, but it wasn't until the early 1960's, that the plastic jug was introduced. This cheaper, lighter, and non-breakable packaging alternative reduced transportation costs, provided safety to the workers involved in shipping and handling, and also prohibited ultraviolet (UV) light from reaching the bleach, which improved its chemical stability and effectiveness. However, in recent years this plastic became an environmental issue, so now the amount of plastic used to create the packaging is reduced, and more recycled plastic is used ("How Bleach is Made", 2007).
This video shows how bleach is filled and capped into plastic bottles.