Introduction to bleach
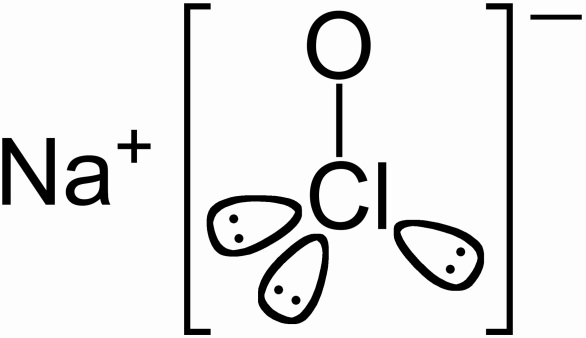
The scientific name for bleach is sodium hypochlorite. It's chemical formula is NaClO, and each element has only one atom.
Other names for sodium hypochlorite are:
- Antiformin
- Carrel-Dakin's Solution
- Chlorine Bleach
- Chloros
- Chlorox
- Dakin's Solution
- Liquid Bleach
- Javax
- Javelle Water
Other names for sodium hypochlorite are:
- Antiformin
- Carrel-Dakin's Solution
- Chlorine Bleach
- Chloros
- Chlorox
- Dakin's Solution
- Liquid Bleach
- Javax
- Javelle Water
Properties of Bleach
Bleach is a clear, slightly yellow liquid, with a characteristic odour. It has a density of about 1g/mL. Bleach is extremely reactive and can cause harsh damages when mixed with certain compunds or chemicals. When bleach conatains 5% sodium hypochlorite, it has a pH of 11, making it irritating, but when it has a concentration of 10-15%, it becomes corrosive. Sodium hypochlorite is also unstable, and disintegrates when heated. This also occurs when it come into contact with acids, sunlight, certain metals and poisonous and corrosive gasses, including chlorine gas. It also reacts with flammable compunds, and is inflammable. It kills bacteria and turns materials white, or lighter (Disinfectants Sodium Hypochlorite, n.d).
Bleach is a clear, slightly yellow liquid, with a characteristic odour. It has a density of about 1g/mL. Bleach is extremely reactive and can cause harsh damages when mixed with certain compunds or chemicals. When bleach conatains 5% sodium hypochlorite, it has a pH of 11, making it irritating, but when it has a concentration of 10-15%, it becomes corrosive. Sodium hypochlorite is also unstable, and disintegrates when heated. This also occurs when it come into contact with acids, sunlight, certain metals and poisonous and corrosive gasses, including chlorine gas. It also reacts with flammable compunds, and is inflammable. It kills bacteria and turns materials white, or lighter (Disinfectants Sodium Hypochlorite, n.d).